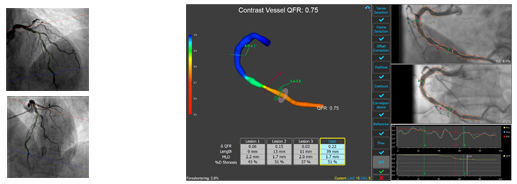
QAngio XA 3D is software intended to be used for performing calculations in X-ray angiographic images of the coronary arteries. QAngio XA 3D enables interventional cardiologists and researchers to obtain quantifications of one or more lesions in the analyzed coronary vessel segment. In particular, QAngio XA 3D provides:
QAngio XA 3D is indicated for use in clinical settings where validated and reproducible quantified results are needed to support the assessment of coronary vessels in X-ray angiographic images, for use on individual patients with coronary artery disease. When the quantified results provided by QAngio XA 3D are used in a clinical setting on X-ray images of an individual patient, the results are only intended for use by the responsible clinicians.
QFR measurements have not been evaluated for non-coronary arteries, pediatric patients, and cardiac patients with the following conditions:
QAngio XA 3D must be used by cardiologists or trained technicians who are qualified to perform cardiac analysis. If the analysis results are used to reach a diagnosis, the results must be interpreted by a qualified medical professional. In clinical practice QAngio XA 3D should not be used for purposes other than those indicated in the section Intended Use.
Users must have sufficient proficiency in the selected operating language, read this manual, become familiar with the software, and must be certified by Medis before using QAngio XA 3D in a clinical environment in order to obtain reliable analysis results.
The shapes of objects and calipers displayed may get slightly distorted when the resolution is set to an aspect ratio different than the monitor’s physical aspect ratio. This distortion does NOT affect the accuracy of measurements or analyses. To avoid distortion, set the resolution of the monitor to an aspect ratio equal to the physical aspect ratio. LCD monitors typically operate best at their native resolution. Microsoft Windows recommends a resolution when it has sufficient information to do so.
QAngio XA 3D has been cleared for market in the United States by the FDA (Food and Drug Administration) under the provisions of Section 510(k) of the Food, Drug, and Cosmetic Act.